
OUR RESEARCH

The Biological Foundation: What Are Telomeres and Why They Matter
Every human chromosome ends with a repetitive DNA sequence known as a telomere. Think of a telomere as a cap on a shoelace—without it, the lace frays and breaks. In cells, telomeres protect DNA from damage during replication. Each time a cell divides, its telomeres shorten slightly. Over time, when they become too short, the cell can no longer replicate properly, entering a state called senescence or undergoing programmed death.
This gradual loss of telomere length acts as a molecular clock for aging. In healthy individuals, that’s a slow, evolutionary process. However, in certain diseases, telomeres erode much faster than normal, leading to premature aging syndromes, tissue failure, and systemic disease. Our company’s mission is to intervene in this process by helping cells maintain telomere stability: repairing the molecular timer that otherwise drives aging forward.
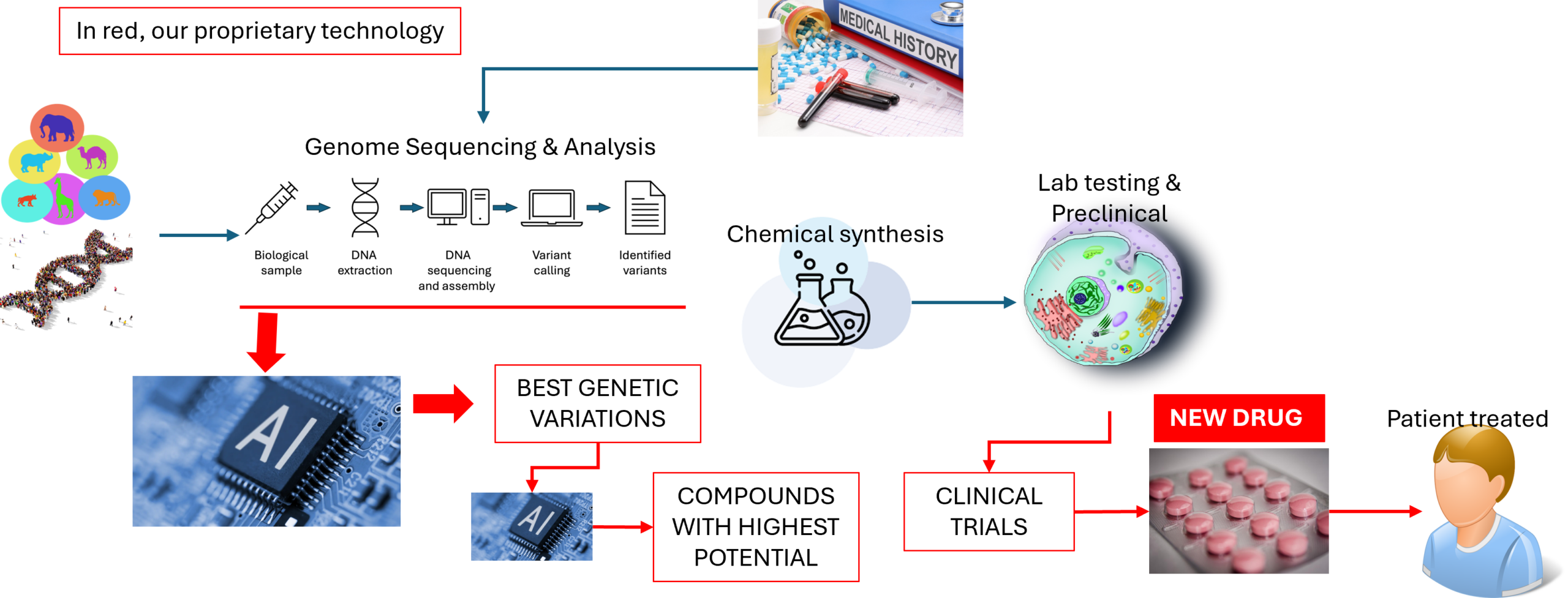
Decoding Nature’s Clues: Studying Telomere-Regulating Genes across Species
The natural world offers profound lessons about longevity. Some species—such as certain whales, bats, or even small rodents—have extreme life spans relative to their size. Many of these animals show a unique pattern of telomere maintenance, hinting at genetic variants that preserve chromosome stability well beyond the human equivalent.
Our research team began by precisely analyzing gene sequences that regulate telomere length and maintenance in many organisms, including humans. Two categories of genetic components proved particularly vital: telomerase and the shelterin complex.
- Telomerase is an enzyme that extends telomere DNA, essentially rebuilding the protective ends of chromosomes. In most adult human cells, telomerase activity is extremely low or absent. This helps protect against unchecked cell division (such as in cancers) but also limits regenerative capacity.
- The shelterin complex is a set of proteins that bind directly to telomeres. It shields them from being mistaken as damaged DNA and controls their length maintenance.
By comparing telomerase and shelterin gene sequences across various organisms, our scientists identified patterns correlating with species longevity and average telomere stability. These comparisons revealed specific genetic variants and protein structures that are associated with longevity and cellular endurance. Using this knowledge, our proprietary AI platform uncovers promising genetic variations that could restore telomere stability in human cells suffering from pathological shortening.

Our Proprietary AI Platform: The Engine of Discovery
Modern biology produces staggering volumes of data. Sequencing genomes across species generates terabytes of information that traditional analysis methods struggle to handle. To interpret and extract meaning from this complexity, we built an advanced artificial intelligence framework specifically designed for telomere biology and longevity research.
This system conducts several operations simultaneously:
- Cross-Species Genomic Mapping:
It aligns and compares telomerase and shelterin gene sequences across dozens of organisms—from short-lived mammals to long-lived vertebrates—and identifies conserved or advantageous sequence variations. - AI-Powered Life Span Correlation:
Through supervised learning models, the platform quantifies how specific sequence variations correlate with average life span, cellular replication capacity, and telomere length across species. - Variant Scoring and Predictive Modeling:
Machine learning algorithms score each variation according to its predicted stabilizing effect on telomere integrity in human cells. The system doesn’t just compare raw sequences—it predicts functional consequences on protein folding, binding affinity, catalytic activity, and overall genomic resilience.
